Drug sold on Amazon recalled. ‘Hidden drug ingredients’ can cause death and diarrhea
One lot of a drug sold as a “dietary supplement” has been recalled after the FDA confirmed it has two undeclared “hidden ingredients” that can cause death, internal bleeding, diarrhea and other problems.
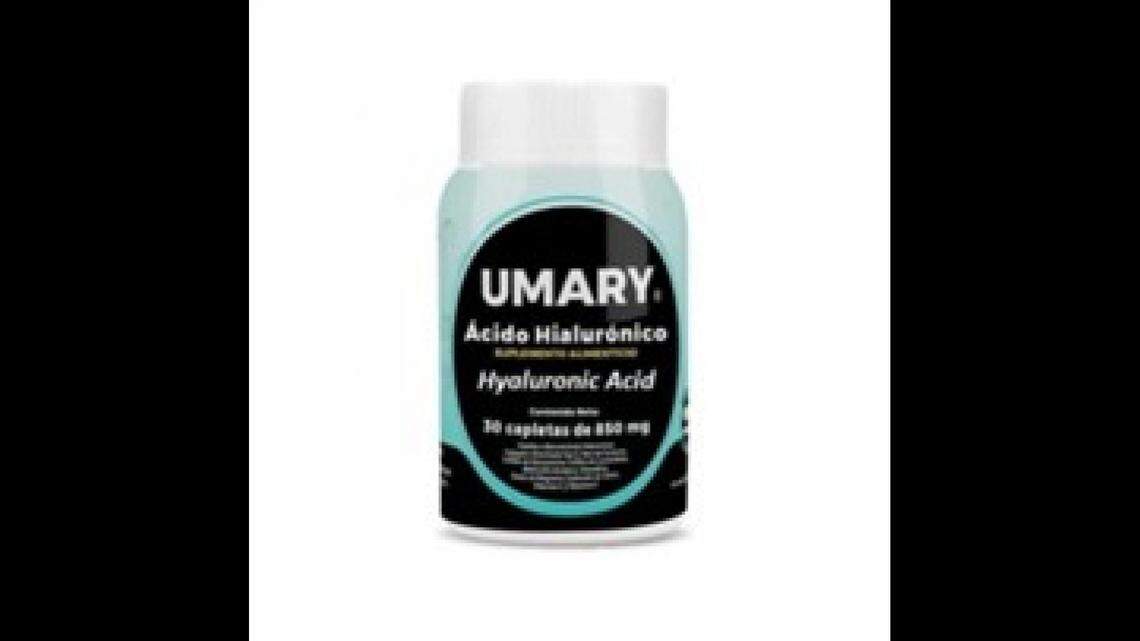
MXBBB sells Umary Acid Hyluronic, 850 mg, in 30-count bottles on Amazon.com and recalled lot No. 24183 with the expiration date of 07/01/28. FDA testing said the Umary has diclofenac and omeprazole, something the packaging doesn’t tell users.
According to the risk statement in MXBBB’s FDA-posted recall notice, diclofenac is a non-steroidal anti-inflammatory drug (NSAIDs) that can increase the risk of “heart attack and stroke, as well as serious gastrointestinal damage, including bleeding, ulceration, and fatal perforation of the stomach and intestines.”
Also, it can mix badly with prescription drugs or other supplements that have NSAIDs.
“Omeprazole is a proton pump inhibitor (PPI) used to treat gastric acid-related disorders,” the risk statement continued. “PPI medicines may cause serious skin reactions, abdominal pain, diarrhea, nausea, and headaches. This hidden ingredient may mask stomach issues such as erosion, ulcers, and stomach cancer” and can mix poorly with other drugs”.
Return the Umary for a full refund. If you have a medical problem, contact a medical professional. Then, contact the FDA’s MedWatch adverse event reporting system, either online or by calling 800-332-1088 to request a reporting form that can be faxed back to the agency.
Consumers with questions regarding this recall can contact MXBBB by emailing Joshua Montoya at Joshua980810@outlook.com seven days a week, 24-hours a day. The phone number for Montoya, 915-258-6936, goes to a voice mail that was full as of Sunday afternoon.
This story was originally published November 24, 2024 at 10:35 AM with the headline "Drug sold on Amazon recalled. ‘Hidden drug ingredients’ can cause death and diarrhea."
