Soaps have been recalled for a contamination that can cause deadly sepsis
Four over-the-counter soaps designed to clean various body areas might contain the microbial contamination that can cause sepsis.
The risk statement in DermaRite Industries’ FDA-posted recall notice for PeriGiene, KleenFoam, DermaSarra and DermaKleen said: “In healthy individuals with minor skin lesions the use of the products will more likely result in local infections, whereas in immunocompromised individuals the infection is more likely to spread into blood stream leading to life-threatening sepsis.”
The issue involves possible burkholderia cepacia microbial contamination.
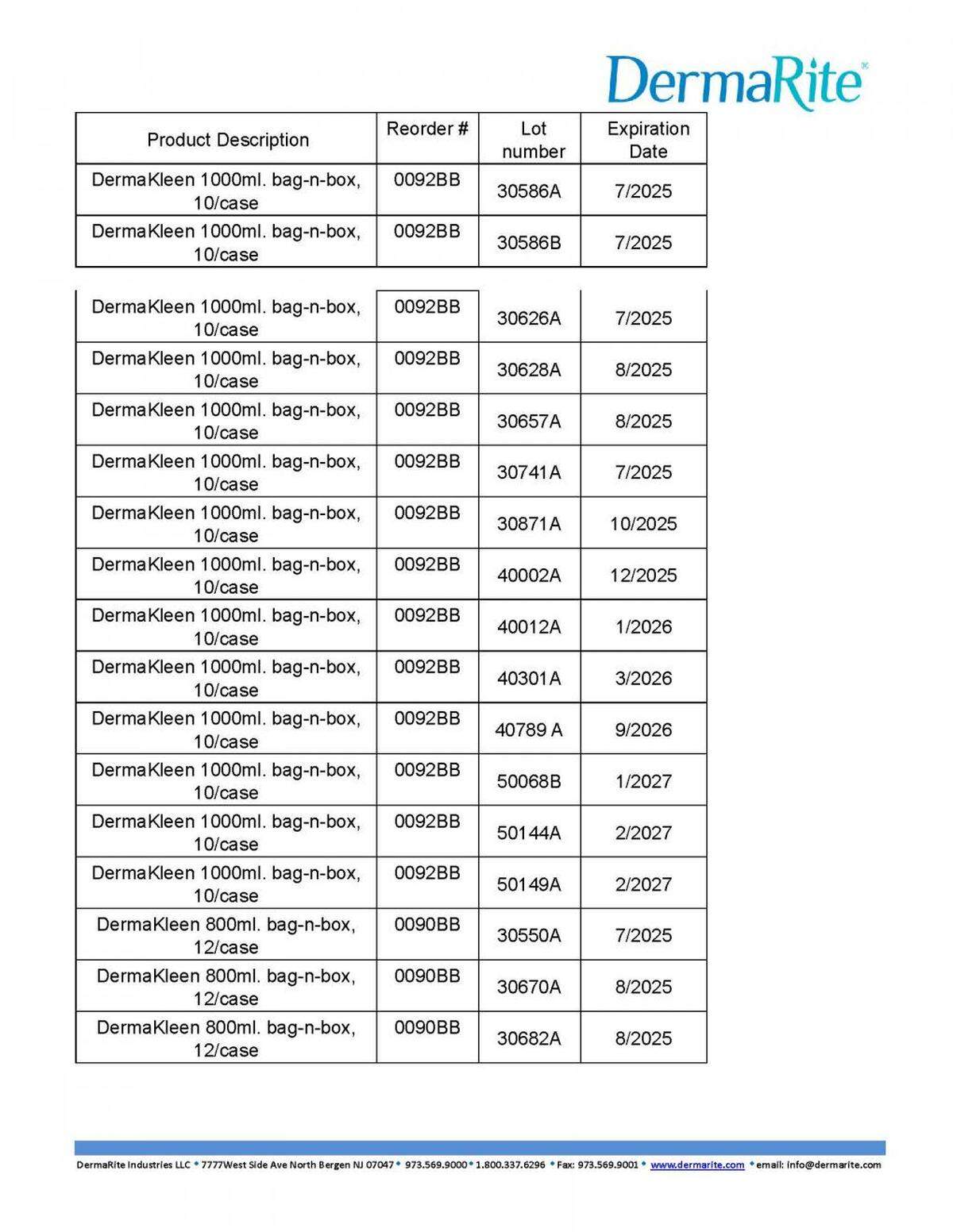
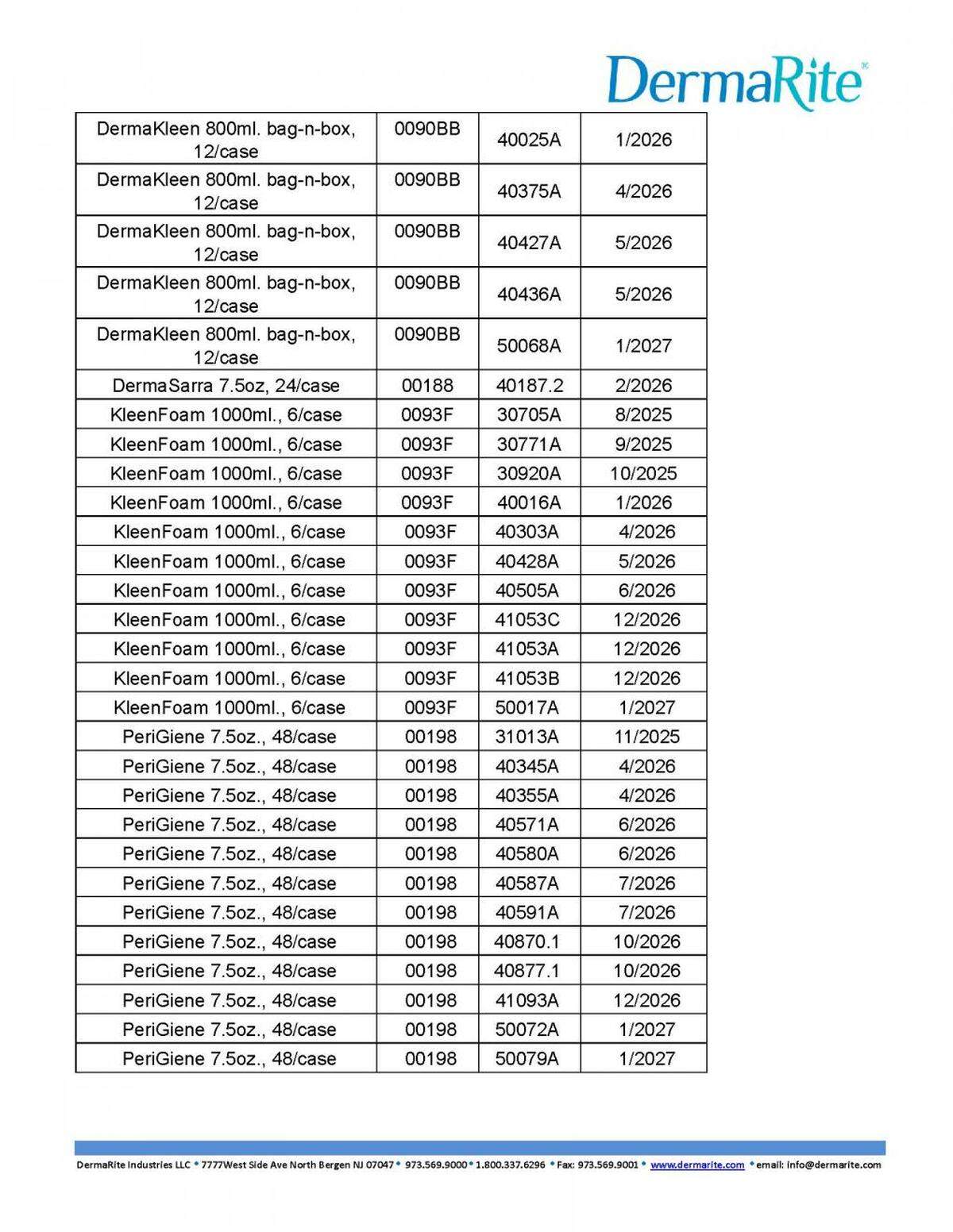
Here’s what’s recalled in the 50 states and Puerto Rico:
KleenFoam, an animicrobial foam soap with aloe vera, is for washing hands before and after dealing with someone under medical care or changing diapers.

DermaSerra is an “external analgesic for temporary relief of itching asosciated with minor skin irritations” from sunburn, insect bites, dry skin or detergents.

DermaKleen is an antiseptic lotion soap with Vitamin E.

PeriGiene is an “antiseptic cleanser” for use in the perineal area, which the Cleveland Clinic identifies as “the tiny patch of sensitive skin between your genitals (vaginal opening or scrotum) and anus, and it’s also the bottom region of your pelvic cavity.”

Look below or at the recall notice for the lot numbers of each recalled.
Questions about the recall should be directed to DermaRite’s Mary Goldberg, 973-569-9000, ext. 104, Monday through Friday, 9 a.m. to 5 p.m., Eastern time, or at email address voluntary.action@dermarite.com.
If you have a medical problem while using this product, contact a medical professional. Then, contact the FDA’s MedWatch program, either online or via a form available by calling 800-332-1088.
This story was originally published August 14, 2025 at 8:16 AM with the headline "Soaps have been recalled for a contamination that can cause deadly sepsis."
